
Publication planning is more than publishing a pivotal manuscript and considering the job done. A strong plan ensures your science reaches the right audiences, at the right time, in the right way, so more people engage with the data and ultimately help achieve the best possible outcomes for patients.
At JB Ashtin, we help pharma and biotech teams move beyond a “paper published, box checked” mindset. The best strategies are proactive, integrated, and designed to amplify impact.
Here are 4 ways we help clients raise the bar
1. Plain Language Summaries of Publication (PLSPs) Increase Accessibility
PLSPs are becoming more of an expectation than a “nice-to-have.” The intent of PLSPs is to make research accessible to non-specialists, including patients and caregivers, and reinforce transparency.
We recommend building PLSPs into the publication plan from the start. An effective approach to developing PLSPs is to collaborate with the original publication’s authors and patient reviewers. This is a transparent approach that gives the PLSP credibility. One of our recent PLSPs translated a complex study involving over 18,000 children into accessible language for caregivers and patients. This work was a collaborative effort between clinicians, patient advocates, and our internal team, out the full summary here.
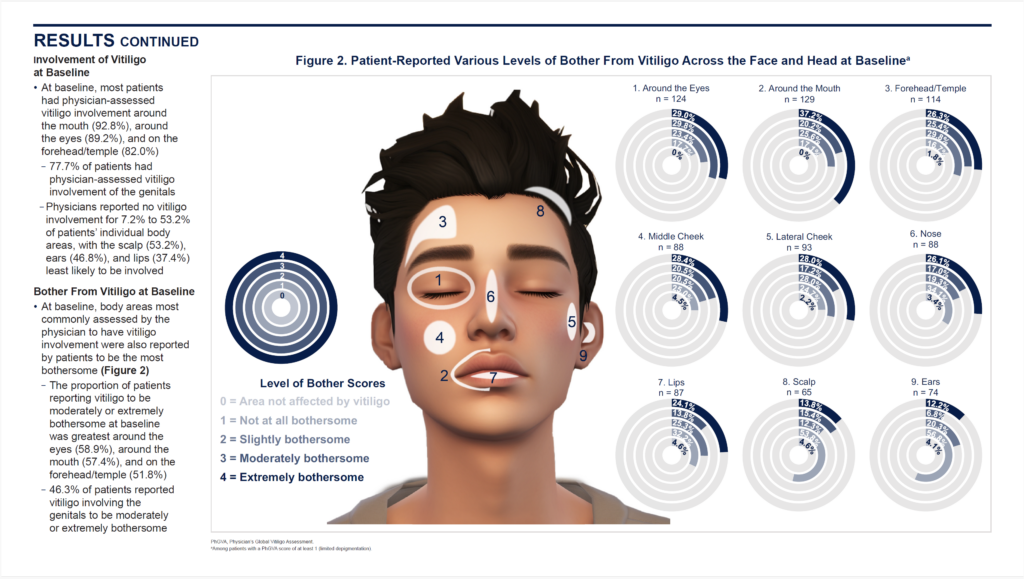
2. Data Visualizations That Tell a Story
Complex data need more than just a table to make an impact. Consider developing graphical abstracts, infographics, and interactive figures to turn complex science into something memorable and shareable. These visual overviews help clinicians, patients, and advocacy groups quickly grasp key results. The old adage, “a picture is worth a thousand words” is indeed true.
3. Extending the Life of Your Publication
Publishing the findings of a clinical trial is just the beginning of stakeholder education. The most effective publication teams plan for how the data will live beyond the journal through interactive posters, videos, podcasts, and other digital channels. This ensures the findings continue to reach HCPs who can act on the information to improve patient care.
Turning a single manuscript into multiple high-engagement assets can broaden reach, deepen engagement, and create more opportunities to connect science to care. There is great opportunity that outweighs any budget concerns of publication teams. Cross-functional alignment and early planning are key to success.
Publishing the findings of a clinical trial is just the beginning of stakeholder education. The most effective publication teams plan for how the data will live beyond the journal through interactive posters, videos, podcasts, and other digital channels. This ensures the findings continue to reach HCPs who can act on the information to improve patient care.
Turning a single manuscript into multiple high-engagement assets can broaden reach, deepen engagement, and create more opportunities to connect science to care. There is great opportunity that outweighs any budget concerns of publication teams. Cross-functional alignment and early planning are key to success.
3. Extending the Life of Your Publication
Publishing the findings of a clinical trial is just the beginning of stakeholder education. The most effective publication teams plan for how the data will live beyond the journal through interactive posters, videos, podcasts, and other digital channels. This ensures the findings continue to reach HCPs who can act on the information to improve patient care.
Turning a single manuscript into multiple high-engagement assets can broaden reach, deepen engagement, and create more opportunities to connect science to care. There is great opportunity that outweighs any budget concerns of publication teams. Cross-functional alignment and early planning are key to success.
4. Using AI with the Right Oversight
AI-assisted writing tools can potentially decrease the time to generate a first draft, speed up the time to identify gaps, and ensure consistency across large, multi-author projects. Theoretically, using AI tools can improve efficiency so we get information into the hands of those who can use it sooner, ultimately supporting faster improvements in patient care. In reality, we are not there yet. There remain concerns about accuracy, privacy, and the need for human judgment and oversight that prevent us from using AI exclusively in the manuscript development process. However, at JB Ashtin, we integrate AI into the brainstorming step: identifying gaps, suggesting interesting titles, and streamlining our research process, without jeopardizing our clients’ confidential data. This allows our writers to focus on scientific interpretation, clarity, and compliance.
Publication planning is more than publishing a pivotal manuscript and considering the job done. A strong plan ensures your science reaches the right audiences, at the right time, in the right way, so more people engage with the data and ultimately help achieve the best possible outcomes for patients.
Let's Build What's Next, Together
We know how crucial it is to get it right in medical communications. If you’re looking for a partner who can balance creativity with compliance and deliver impactful results –
